FDA Pulls Impella Purge Cassettes After Device Failures
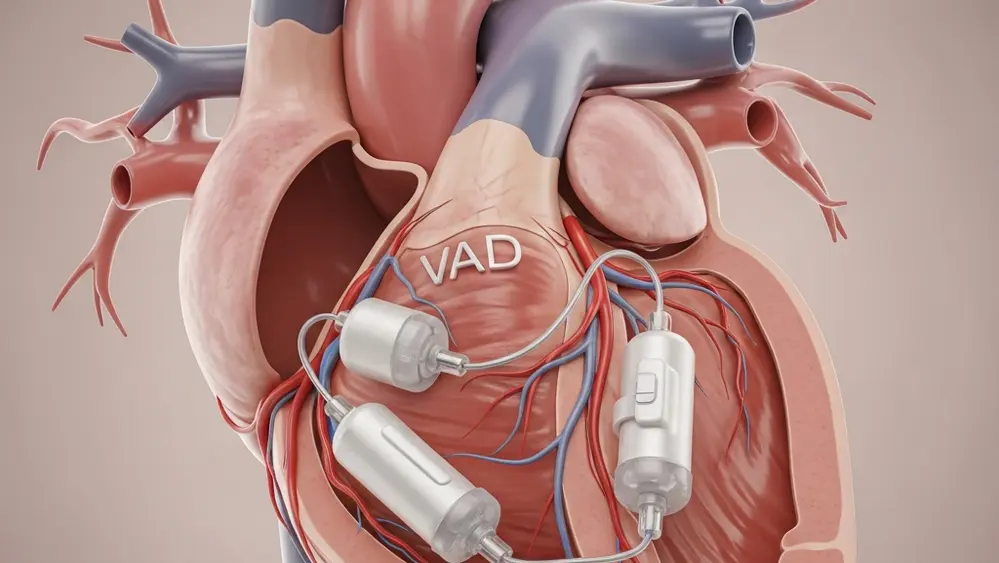
The FDA recently issued a high-priority notice regarding the removal of Impella Purge Cassettes from clinical use following reports of critical system failures. According to recent reports, these devices, which are essential for maintaining heart pump functionality, have experienced issues that lead to the loss of circulatory support. Patients relying on these pumps for life-sustaining cardiovascular assistance are now facing increased risks of serious injury or death. Abiomed, the manufacturer, has initiated a voluntary removal to address these mechanical defects before more patients are harmed. This development highlights a significant safety concern for thousands of cardiac patients currently undergoing treatment in hospitals nationwide. The failure of these cassettes can cause the entire heart pump system to cease functioning without warning.
Affected by a Product Liability Issue?
Our specialized tool can help you estimate the potential worth of your case based on current laws and precedents.
Holding Manufacturers Accountable for Defective Heart Pumps
From a legal perspective, the liability for these failures often falls on the manufacturer under the doctrine of strict product liability. When a medical device fails to perform its intended function or contains a design defect that makes it unreasonably dangerous, the company may be held responsible for the resulting damages. Attorneys will investigate whether the manufacturer knew about these risks and failed to warn healthcare providers in a timely manner. In cases involving such critical life-support equipment, the burden of ensuring device integrity is exceptionally high. If a device failure leads to a fatality, the family may need to determine who can file a wrongful death lawsuit to seek justice for their loss. Companies must be held to account when their products fail those who need them most.
Critical Steps for Patients Impacted by Device Recalls
If you or a loved one has been treated with an Impella heart pump, it is vital to monitor for any signs of device malfunction or decreased circulatory efficiency. You should consult with your medical team immediately to confirm if the specific purge cassettes used in your care were part of the affected lot. Documenting the timeline of the device's use and any subsequent health complications is a necessary step in building a potential legal claim. Families dealing with the aftermath of a device failure should also understand how conscious pain and suffering impacts the legal value of a survival action. Seeking a comprehensive medical evaluation can provide the necessary evidence to link the device failure to the patient's current health status and eventual recovery path.
Understanding Potential Damages in Medical Device Claims
Victims of defective medical devices may be eligible for significant compensation covering a variety of economic and non-economic losses. This often includes medical expenses for corrective surgeries, long-term care needs, and lost wages due to extended recovery periods or permanent disability. In the most tragic circumstances, legal teams must also consider how dependents and wrongful death calculations will factor into the final settlement or verdict. Pain and suffering, along with loss of consortium for family members, represent substantial portions of these claims. Settlement values are highly individualized and depend on the severity of the injury and the degree of negligence proven against the manufacturer during the litigation process.
Regulatory Oversight and Product Liability Statutes
The legal framework for medical device litigation is governed by both federal regulations and state-specific product liability laws. The FDA classifies such recalls under strict categories, often labeling them as Class I if there is a reasonable probability of serious adverse health consequences. Consumers should be aware that the FDA Medical Device Reporting (MDR) system is a primary tool for tracking these failures. Furthermore, the Legal Information Institute provides a detailed overview of how product liability theories apply to manufacturing defects. Statutes of limitations vary by state, meaning there is a limited window of time to file a lawsuit after an injury is discovered. Understanding these deadlines is essential for preserving your right to financial recovery and ensuring your voice is heard in court.
Calculate Your Potential Case Value Today
Navigating the complexities of a medical device lawsuit can be overwhelming, especially when dealing with the physical and emotional toll of a heart condition. Our team is dedicated to helping victims understand their rights and the potential value of their claims through our specialized tools. By using our free case evaluator, you can get a clearer picture of what your recovery might look like based on the specifics of your situation. Do not leave your financial future to chance after a manufacturer’s error has put your life at risk. Take the first step toward securing the compensation you deserve by clicking through to our calculator today. We are here to provide the clarity and support you need during this challenging time.
Disclaimer: This blog post is for informational purposes only and does not constitute legal advice. For specific legal guidance regarding your situation, please consult with a qualified attorney.